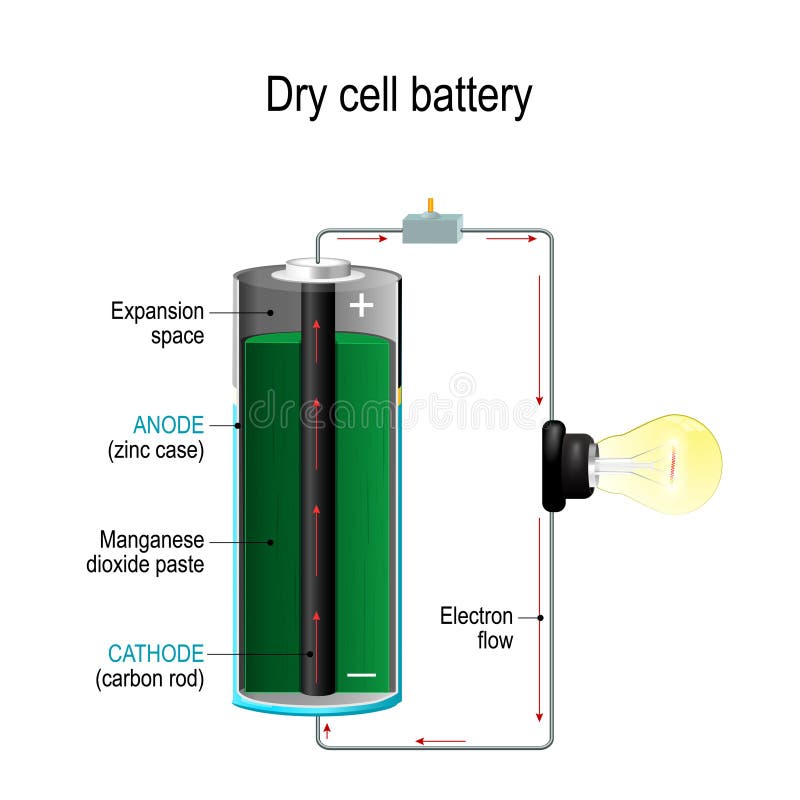
PJP Eye also touts the possibility of improving battery performance as well as making batteries greener. But there have been reports of dangerous working conditions there.įrom seawater to biowaste and natural pigments, there is a long list of potential alternatives in nature that would be much more widely available – the hard part is proving that any of them can realistically compete with the kinds of batteries already on the market, which are seemingly so indispensable in our gadget-strewn world. To take another example: cobalt, which is used in many lithium-ion batteries, is predominantly extracted in the Democratic Republic of Congo. "It's very easy to imagine, as a battery material goes through mining and transportation, how that carbon footprint can really add up," says Sam Wilkinson, an analyst at S&P Global Commodity Insights. Graphite, too, is mined or made from fossil fuels, both of which also have negative environmental impacts. The recovered lithium is often shipped long distances from where it is mined to be refined in countries such as China. Extracting the metal requires large amounts of water and energy, and the process can leave huge scars in the landscape. Lithium mining can have a considerable impact on the environment. Like PJP Eye, they argue we could be using much more sustainable and widely available materials for battery production. With huge demand for batteries expected in the coming years, propelled by the rise of electric vehicles and large energy storage systems, some researchers and businesses are frantically developing possible alternatives to the lithium ion and graphite batteries that are commonplace today. The majority of batteries use graphite as an anode but PJP Eye argues their approach is more sustainable, since they can make anodes using waste cotton from the textile industry. Ions move in one direction when the battery is charging and in the other direction when it releases energy to a device. In the batteries developed by the company, together with researchers at Kyushu University in Fukuoka, Japan, carbon is used for the anode – one of the two electrodes between which flow ions, the charged particles in batteries. The firm bought a shipment of cotton in 2017 and still hasn't used all of it, says Okina. 
And that 1kg (2.2lbs) of cotton yields 200g (7oz) of carbon – with just 2g (0.07oz) needed for each battery cell. Okina does say that a high temperature is required, above 3,000C (5,432F). Pressure is secret," he continues, cagily. "The temperature is secret and atmosphere is secret. "The exact process is secret, to be honest with you," says Inketsu Okina, chief intelligence officer at PJP Eye, the Japanese firm that made the battery. For this cash machine has a backup battery inside it – a battery that contains carbon from carefully combusted cotton. But on a street in India, there's a cash machine still happily dispensing banknotes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
